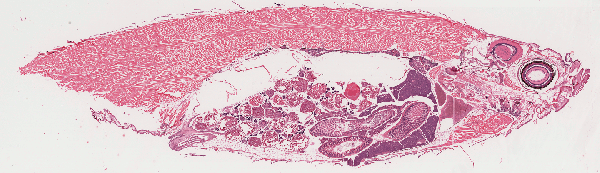
 Fixed fish are processed for histological sectioning and staining with hematoxylin
and eosin. Additional special stains may be used to identify infectious organisms.
Histopathology allows for evaluation of all tissues for normal organ development,
presence of infectious agents, responses to suboptimal water quality, and a range of
tissue changes and pathologies. We, therefore, recommend histopathology for
routine health screening of sentinel and sick fish, and additional diagnostic tests are
provided as appropriate based on histological findings.
Fixed fish are processed for histological sectioning and staining with hematoxylin
and eosin. Additional special stains may be used to identify infectious organisms.
Histopathology allows for evaluation of all tissues for normal organ development,
presence of infectious agents, responses to suboptimal water quality, and a range of
tissue changes and pathologies. We, therefore, recommend histopathology for
routine health screening of sentinel and sick fish, and additional diagnostic tests are
provided as appropriate based on histological findings.
 We perform aseptic cultures of the kidney and swim bladder for diagnosis of
systemic infections and bacterial aerocystitis, respectively. Samples are streaked on
blood agar plates. Fish are shipped live for bacteriology, although samples can be
taken from frozen specimens if
necessary. We require advanced
notification prior to shipment of live
fish to ensure appropriate personnel
and lab supplies are available.
Bacterial infections in zebrafish are
often sporadic and opportunistic. We
recommend bacteriology as a follow-up
to histopathology if bacterial infections
are observed in tissue sections and in
conjunction with histopathology
during an incidence of increased
morbidity or mortality.
We perform aseptic cultures of the kidney and swim bladder for diagnosis of
systemic infections and bacterial aerocystitis, respectively. Samples are streaked on
blood agar plates. Fish are shipped live for bacteriology, although samples can be
taken from frozen specimens if
necessary. We require advanced
notification prior to shipment of live
fish to ensure appropriate personnel
and lab supplies are available.
Bacterial infections in zebrafish are
often sporadic and opportunistic. We
recommend bacteriology as a follow-up
to histopathology if bacterial infections
are observed in tissue sections and in
conjunction with histopathology
during an incidence of increased
morbidity or mortality.
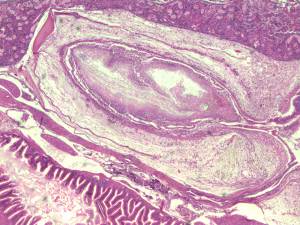 Bacterial culture and histopathology can be performed on the same fish. We use a
dorsal approach to perform aseptic cultures of the kidney and swim bladder. After
samples are taken for cultures, the carcasses are fixed in Dietrich’s fixative and
processed for histological sectioning and H&E staining. Interpretation of culture
results is greatly enhanced by the addition of histological evaluation on the same
fish. By also examining tissue sections for evidence of bacterial infection, we
mitigate the risks of false culture results. False negative culture results may occur
with improper sampling of focal infections and with slow-growing and difficult to
culture bacteria. False positive
cultures may occur with skin or gut
contamination of samples, which is
common with small fish, and in
mixed bacterial infections where
the easily cultured bacteria are not
responsible for the observed fish
morbidity and mortality. By
adding histopathology we also have
the opportunity to diagnose
numerous other pathologies not
associated with bacterial infection.
Bacterial culture and histopathology can be performed on the same fish. We use a
dorsal approach to perform aseptic cultures of the kidney and swim bladder. After
samples are taken for cultures, the carcasses are fixed in Dietrich’s fixative and
processed for histological sectioning and H&E staining. Interpretation of culture
results is greatly enhanced by the addition of histological evaluation on the same
fish. By also examining tissue sections for evidence of bacterial infection, we
mitigate the risks of false culture results. False negative culture results may occur
with improper sampling of focal infections and with slow-growing and difficult to
culture bacteria. False positive
cultures may occur with skin or gut
contamination of samples, which is
common with small fish, and in
mixed bacterial infections where
the easily cultured bacteria are not
responsible for the observed fish
morbidity and mortality. By
adding histopathology we also have
the opportunity to diagnose
numerous other pathologies not
associated with bacterial infection.
Fish should be shipped live for necropsy examination. Fish are euthanized,
dissected, and organs examined for gross changes. Fin clips, gill clips, and skin
scrapes are also performed. Tissues may be fixed and processed for histological
sectioning and staining.
Real-time PCR assays for diagnosis of Pseudoloma neurophilia, Mycobacterium chelonae, M. haemophilum, and M. marinum are performed on frozen and paraffin- embedded tissue at the Oregon Veterinary Diagnostic Laboratory. PCR assays for additional zebrafish pathogens, such as Pseudocapillaria tomentosa, will be available soon. Samples are received by ZIRC and transferred to OVDL for appropriate testing.
Diagnosis of mycobacteriosis and species identification of Mycobacterium spp. require different approaches compared to Gram negative bacteria. Diagnosis of mycobacteriosis to the genus levels only requires observation of acid-fast bacteria in infected tissues. Species identification usually requires DNA sequence evaluation (e.g., 16S rDNA or hsp65) or even HPLC comparisons of mycolic acids from cultured bacteria. We use the former. For severe outbreaks, we often compare the DNA finger prints of various isolates using molecular techniques (Ostland et al. 2008; Whipps et al. 2008). Several methods can be employed to identify mycobacteria in zebrafish. As histopathology is our primary diagnostic method, mycobacteriosis is usually first identified by observing lesions consistent with the disease (e.g., granulomas) in tissue sections. For confirmation, we then follow with acid fast staining of additional sections from the same fish. Diagnosis to the genus level can also be accomplished by staining and identification of acid fast bacilli in tissue imprints. Following diagnosis, different options are available for species identification.
Whereas Mycobacterium spp. generally grow slower in culture than Gram negative bacteria, their resistance to many antibiotics and disinfectants such as cetyl-pyridinium chloride (CPC) allows for selective culture on specific media. Here organs such as liver, spleen and kidney are aseptically cultured, mycobacteria are isolated on media, and then identified using molecular techniques.
Some mycobacteria, such as M. haemophilum, are extremely fastidious and slow growers. Therefore, we often find it more appropriate to conduct PCR analysis directly on infected tissues (Whipps et al. 2007). When Mycobacterium infection is suspected, spleen can be removed and frozen back before the fish carcass is preserved in fixative and processed for histopathology. PCR analysis can then be performed on the spleen if acid fast bacilli are observed in histological sections.
Molecular diagnosis of mycobacteria in zebrafish in which the fish have been preserved in formalin-based fixatives and processed into paraffin can be obtained with reasonable success (Peterson et al. 2013). Paraffin blocks containing fish identified as positive by histopathology are sent to the Veterinary Diagnostic Laboratory (VDL) at Oregon State University where infected fishes are cored from blocks and evaluated with specific PCR tests for M. marinum, M. chelonae and M. haemophilum, the three most common species causing mycobacteriosis in zebrafish (http://vetmed.oregonstate.edu/diagnostic). The above methods (a-c) are often employed with multiple fish during an outbreak – e.g., some fish preserved in Dietrich’s processed for histology and other fish frozen or preserved in ethanol for PCR analysis. However, we often use an alternative approach in which use a single fish is processed for both histology and other tests (e.g., culture/imprints/direct PCR) as follows: 1) Aseptically expose the coelomic cavity, 2) aseptically obtain biospies of spleen, liver, mesenteries, 3) preserve fish in Dietirich’s for histopathlogy, 4) prepare imprints of biopsied tissues and perform acid fast stains, 5a) freeze biopsy specimens for future PCR analysis or 5b) setup bacterial cultures for mycobacteria using CPC disinfection followed by culture on mycobacteria-specific media. With this approach, we can correlate bacteriology results with specific severities of infection. Alternatively, we can process several fish for histology, and then select a subset for further bacterial identification based on histology results.
Ostland, V.E., Watral, V., Whipps, C.M., Austin, F.W., St-Hilaire, S., Westerman, M.E., Kent, M.L. 2008. Biochemical, molecular, and virulence characteristics of select Mycobacterium marinum isolates in hybrid striped bass (Morone chrysops x Morone saxatilis) and zebrafish (Danio rerio). Dis. Aquat. Org. 79: 107-118. Peterson, T.S, Kent, M.L. Ferguson, J.A., Watral, V.G., Whipps, C.M. 2013. Comparison of fixatives and fixation time on PCR detection of Mycobacterium in zebrafish Danio rerio. Dis. Aquat. Org. 104:113-120. Whipps, C.M. Dougan, S.T., Kent, M.L. 2007. Mycobacterium haemophilum infections of zebrafish (Danio rerio) in research facilities. FEMS Microbiol Lett. 270: 21–26. Whipps, C.M., Matthews, J.L., Kent, M.L. 2008. Distribution and genetic characterization of Mycobacterium chelonae in laboratory zebrafish (Danio rerio). Dis. Aquat. Org. 82: 45-54.